My thoughts about the fuel E85 compared to other ethanol products which not had gone through an expensive chemical dehydration process, have led me into this experiment. The aim is to ascertain how much water it can absorbed without gasoline starts secede. Furthermore, this has developed into a self-composed method, for from an arbitrary sample of E85 determine its potential water content.
I think I need to clarify what this is about first:
As I said, to get ride of the remaining water need the ethanol that shall be blended into E85 to be going through a costly process. It is only possible to distil alcohol up to 96%. The remaining water can not be distilled off, but must via the chemical way be eliminated from ethanol. The purpose of doing this is controversial. Many believe that it is due to ethanol that contain for about 5% water, not have the capability to be mixed with gasoline. What I've found when I measured the fuel consumption was that ethanol/E85 as a fuel that containing water, becomes less energy-rich (the mileage will drop). Its fine to tank in a car but it gives a smaller energy gain. Contrary to popular belief goes it very well to mix large quantities of water in E85, without that ethanol and the gasoline separates from each other. This is a bit worrying, because them who selling E85 can mix water (at most 20%) and then get charge as it was E85 without water.
In order to illustrate which quantities of water this is, in the case of E85 and its absorptive capacity, have I done three trials. One with E85 and two with commercial alcohols (cleaning and washer fluid). Just these three samples do not account for MTBE content in the unleaded petrol 98 RON. According to a vague indication contains unleaded 98 RON about 10% MTBE. Furthermore, is the water absorption capacity, greater for unleaded 98 RON than for unleaded 95 RON. 95 RON also contain MTBE but the content is up to 20%! Shuffled 15% 95 RON with 85% ethanol, will the MTBE content fall to 3%. The level of gasoline for the three samples is about 1.5 ml lower then. MTBE is an octane enhancing fossil fuel, but is soluble in water/alcohol.
For the samples are distilled water added in 100 ml ethanol fuel. The importation of water is interrupted when the sample shows signs of turbidity. Muddiness indicates that phase separation has begun. Phase separation is the same as the stratification of gasoline and ethanol/water element and depends on the temperature and the proportions of the components. A major component of ethanol provides large water absorption, while a small provides less absorption.
T-red is the name of the red-colored ethanol we use here in Sweden, for cleaning, etc. T-blue is the name of a very similar product as T-red, but it is blue and should be mixed with water for use as washer fluid. There is no need to squeeze out a few per cent of water here, because the aim is not to use it as a fuel, such as E85 is. Therefore, these kinds of ethanol only are manufactured through a distillation process, which is cheaper.
| T-red | T-blue | E85 |
| 85 ml T-red and 15 ml U98 | 85 ml T-blue and 15 ml U98 | 98 ml E85 and 2 ml U98 |
| Amount of water = 25 ml | Amount of water = 17 ml | Amount of water = 22 ml |
| Percentage of water = 24% | Percentage of water = 18% | Percentage of water = 22% |
The percentage is the weight of the water quantity, the weight in proportion to the total mass of fuel. The calculations are made on the assumption that the density of ethanol is 0.79 gram/ml and 0.74 gram/ml of gasoline.
Since these are large amounts of water became the change of the proportions of ethanol and gasoline rather drastically. These will therefore be smaller than they were before the water was added.
To investigate how well the absorption of the water-saturated sample was I have to do this temperature test:
It had to endure -18 Celcius (zero Fahrenheit)and also heated to the boiling point.
It showed no signs of separation.
Weight per cent water in the ethanol portion of each sample:
| T-red | T-blue | E85 |
| 73% ethanol & 27% water | 80% ethanol & 20% water | 76% ethanol & 24% water |
New evidence gathered revealed that the same ethanol for T-red also use for production of E85. This is why T-red boarder so little water - it is treated in a dehydration plant. Aside from the fuel stations that can cheat us of money due to hydrated E85 can you with limited resources even calculate how much the water content is of a given E85. I will show some milestones in the calculation of this issue.
According to Statoil's information sheet (no longer exist) is/was the density of E85 784 kg/m3 for summer quality and 782 kg/m3 for winter quality. Transformed to smaller units are the medium density 0.783 g/ml.
100 ml E85 (Statoil) therefore weighs 78.3 grams and can be divided into these components:
- 3 % is MTBE => 2.35 grams
- 85 % is ethanol => 66.56 grams
- 12 % is gasoline => 9.40 grams
When E85 is contaminated of water change the proportions. We have a water component which occupies a certain percentage, how much we do not know. A layer sample shows not the actual content of gasoline and ethanol, if there received water after it was produced. If E85 is diluted with water will the proportion of gasoline and the ethanol get lower than it was from the beginning.
I did a number of experiments with different E-grades. Later could these charts take shape and by using them can we see how much water that can be absorbed before phase separation occurs.
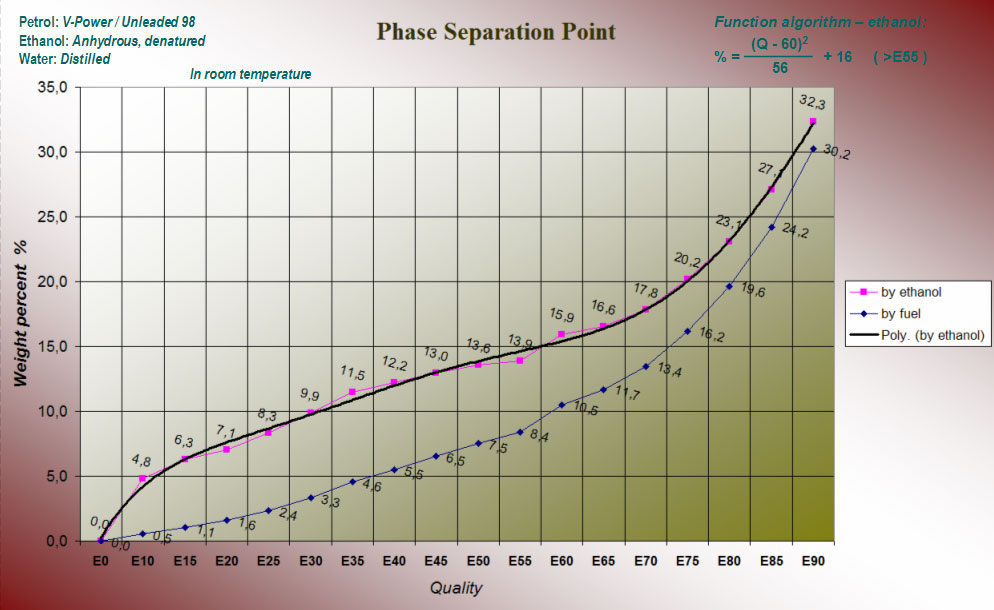
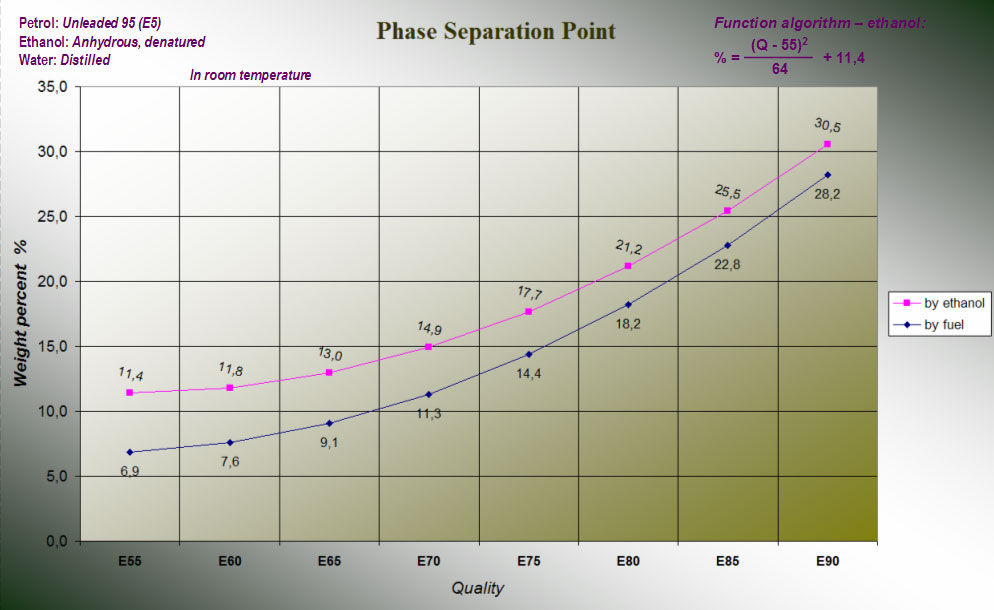
From the graphs one can read the phase separation point in the weight of fuel (by fuel) and in particular the alcohol component (by ethanol) in the fuel. Apparently distinct the phase separation point quite markedly, depending on the type of petrol that is used. If 98 octanes or V-Power is used can the petrol component absorb more water than if it was regular unleaded 95. What this depends on is difficult to predict but one could for example imagine that the finer varieties of petrol are able to absorb more water - unleaded 95 (E5) has already ethanol as an absorbing additive... Since the difference is so obvious, we should be able to decide which sort of petrol it is under a test performance?
The analytical result for the water content will always be based on the algorithm for V-Power/98ron. I have made a wizard in Excel, which can quickly calculate the water content in E85, with the help of a previous layer- and absorptions sample.
Imagine now that a layer samples showed E90, although the sample contains water - for example, 10 % water in ethanol (10 % of water not is equivalent to 10 g of water here). Since the sample contained 10 % water, was it E89. Another example: We have E65, it contains 10 % water - thus it is E63. We have E75 and it contains 10 % water - it is E73, etc. One can then conclude that the difference is marginal in comparison with possible measurement errors. The percentage deviation of the E63 and E65 is 0.6 % - slightly more than half a per cent and for E89/E90 - 1.3 %. But always count on with an error around ± 0.5 % for the lower and ± 1 % for the higher quality (if the content of water is up to 10 %).
We get to work. Measure out 100 ml of E85 and carefully add water while stirring (at room temperature). Stop when the sample shows signs of turbidity. When this happens begin what is called phase separation - the gasoline/alcohol start to separate. Note the amount of water added in millilitres. I assume you have already done a layer sample, if not - determine the quality of a layer sample. For example, you could add 10 ml of water in the E88. - What percentage is it? 10 ml is 10 grams and the weight of ethanol is 88 x 0.783 = 68.9 g.
The addition of water in this sample was: 100 x 10 / (10 + 68.9) = 12.7 % (weight per cent water of the ethanol part)
Algorithm for max-ceiling to V-Power: (88 - 60)2 / 56 + 16 = 30.0 % (maximum weight per cent water in ethanol for E88)
Algorithm for max-ceiling to E5: (88 - 55)2 / 64 + 11.4 = 28.4 % (maximum weight per cent water in ethanol for E88))
The sample contains water because only 12.7 % of water could be added (not 28 or 30). The proportion of water in the alcohol for E88 is (with V-Power as the reference): 30.0 - 12.7 = 17.3 %
To compensate for the error mentioned above, the result should be this (17.3 - 2): 15.3 %
The per cent error is there partly because it is hard to read the test when it becomes cloudy (1 %) and partly because the water content exceeds 10%.
|
| |
|
| |

