SCIENCE →
| by CEO of EE | • 2021-11-15 | |||
| Earth bluish atmosphere | ||||
I often think of different peculiarities concerning the Earth and then try to compare with Venus and Mars (our closest neighbors). The fact that it is more than one celestial body, and that they are crowded in almost the same place makes it easier to study complicated things, such as the way the planets move or rotate, the size of the planet, differences in how the atmosphere is built with regard to electrical properties, magnetism, seasonal changes, distance to the sun, the composition of matter, weather conditions, temperature, color changes, etc, etc. | ||||

IMAGE ©WallpaperSafari
According to the hypothesis I will present here, the blue is due to the existence of Oxygen. However, this hypothesis is not
supported by the established scientific discipline. Thus, that there is no connection between a blue atmosphere and the presence of oxygen. They act completely
independently of each other and the Earth's atmosphere could just as easily be exchanged for any colorless gas, it would mark itself as a blue sky, both from
the ground and from space. The reason that Mars has a red atmosphere is because the thin atmosphere of this planet accommodates iron dust particles.
The atmosphere of Venus is not visible because the entire one is wrapped in a thick layer of clouds.
The physics behind these findings can be difficult for a newbie, but I have also had problems getting to know these theories about what is called “scattering”. By the way, we begin with to first describe the accepted model.
It is the British physicist Lord Rayleigh who gave the name to the phenomenon and who is the reason why light can be scattered - for example in an atmosphere. Rayleigh scattering is a form of weak interaction between photons and electrons, which means that light is produced. When the electrons (bound in the atoms of the air molecules) vibrate in step with the photons of the incoming sunlight, the air molecules (the electrons in the air molecules) begin to emit light. The color of the generated light depends on the size of the particle. A molecule is quite small in comparison with, for example, an iron dust particle. The larger the particle, the redder the light, but if the particle is small (a molecule), blue light is emitted.
This is not about high-energy photons that hit electrons, such as photoelectric absorption or Compton scattering, where a photon strike an electron away. If this happened, it would result in X-rays! In such circumstances, the concept of scattering is very obvious! It radiates in all directions. The closest type of interaction reminiscent of Rayleigh scattering is Thomson scattering.
It is only in certain types of materials that Rayleigh scattering can take place, it is more common for reflection to occur, but sometimes both scattering and reflection occur at the same time. A material condition can here be something that is transparent, such as a liquid, gas, or glassy mineral. For example, a crystal that is bluish. Red and green are also scattered, but blue is scattered most. Overall, the blue color dominates, so it looks blue. You can test it yourself by manipulating the RGB settings that control a color (in a computer program of some kind). Red is given a low value, green slightly larger while blue is given the largest value - the result is blue.
There are three things that can happen when light hits matter:
- light is absorbed by matter
- light is reflected by matter
- the light is neither absorbed nor reflected by matter
If you are a radiation physicist and compare scattering with reflection, you can briefly describe scattering as an interaction between photons and electrons, instead of the photons bouncing away from the electron. So I'm pretty sure this is the basic hypothesis about what is called scattering. That the blue is generated from the air's molecules (oxygen and nitrogen) due to the sunlight interacting with the air atoms' electrons at high altitudes. It is therefore not a requirement that it must be oxygen and nitrogen, because it could just as easily be pure carbon dioxide or any gas, as long as it is not colored. Regardless of gas, it would result in a blue sky, both from the ground and from space.
Then we have the formation of the gas Ozone (trivalent oxygen). Ozone can also be find at high altitudes (the stratosphere), but in contrast to oxygen or nitrogen gas, ozone gas is bluish. Ozone is also created by sunlight, more specifically by ultraviolet light. Apparently, the amounts of ozone are high enough so that harmful levels of UV light not will be able to reach the earth's surface. The UV light from the sun breaks down O2 molecules into simple O1 molecules, which later form O3 molecules. The process requires quite a lot of energy, energy taken from the sun's UV spectra. Because O3 molecules are unstable, they have a short lifespan. It is about hours before an O3 molecule is broken up and regenerated into O2 molecules. Ozone gas is thus a very volatile phenomenon; in addition, ozone is heavier than air and moves downwards until it decomposes into oxygen again. It is thus a process that goes on all the time - oxygen becomes ozone, which becomes oxygen, which becomes ozone, which becomes oxygen, etc. As long as there is oxygen and UV light, there is plenty of ozone. The formation of so-called ozone holes in Antarctica during the winter is due to a lack of UV and winds. It is around the equator that most ozone is formed, and then it is transported to the Polar Regions by the winds. The thickness of the ozone layer is about 30-40 km and the lower limit starts at an altitude of 10-20 km, the same limit where the stratosphere begins. The concentration is 2-8 ppm. If the ozone gas were concentrated to pure ozone and added as a layer at ground level, its thickness would be only a few millimeters. The ozone layer filters out all UVC but partially lets through UVB and all UVA. The human sense of smell is sensitive to ozone gas - the smell of ozone is reminiscent of chlorine. Not even the most expensive measuring instruments are capable of detecting ozone before we do it with our noses. Already 0.1 ppm in the air is an unhealthy content.
However, the light blue color of ozone gas has nothing to do with the blue skies; it is the Rayleigh scattering (within the same atmospheric zone) that is the main cause. The fact that ozone emits blue is due to reflections, not by the interaction between photons and electrons even though ozone is a gas. One obviously has to learn to distinguish between reflection and scattering… Scattering takes place when the gas is colorless, while reflection - the gas is NOT colorless. But if the circumstances allow, both reflection and scattering occur simultaneously.
There are also other oxygen compounds that are bluish. Liquid oxygen is pale blue, the same applies to ordinary water which consists of 89 % oxygen - it is slightly bluish. This is easy to ascertain as the oceans from space are blue, which gives an amplifying effect = atmosphere + oceans. However, heavy water, oxygen, and nitrogen gas as well as liquid nitrogen are colorless.

IMAGE ©ThoughtCo
On cloudy days, both the sky and the water appear gray, but if there is a gap in the cloud cover, you can see the blue sky above. Depending on how dense the clouds are or how much light slips through, the gray varies from white to gray-blue. Really dense clouds (thunderclouds or hurricanes) that are approaching or moving away have a dark blue / dark gray hue seen from the ground. Here one might think that the moisture content is so high that the blue color of the water is again discernible, but from a space point of view, the clouds are always white regardless of its thickness. From the space observatory, there is a big difference in the color of gaseous water and liquid water.
If you undertake a journey with a rocket that starts from the ground towards space, you will not see any of the blue atmospheres as long as you are still in the troposphere and not looking upwards. As you pass through the stratosphere, you should be able to observe a faint blue environment outside the rocket window. When you approach to space and look down, you see through the blue layer in the stratosphere and the ground surface below this has a blue hue. What makes the sky blue is thus about a blue layer that is in the lower part of the stratosphere.
It is said that if the day ends with a red sunset, there is a good chance that the weather will be nice the next day. In other words, it is quite cloudless outside. Why then does the sky turn red when it should be blue then? Here I would like to say that scattering/spreading and/or refraction matters. The high-energy content of the sunlight is consumed before it reaches the eyes of the viewer. Now the sunlight must wrestle with moisture particles and ground dust that resides in the troposphere over a very long distance. This is a completely different thing than when the sunlight comes from above.
How is it now? Is it about Rayleigh scattering or about ozone? You can divide the question into three options.
- the sky is blue due to Rayleigh scattering
- the sky is blue due to ozone (which is a bluish gas)
- the sky is blue due to both Rayleigh scattering and ozone
Someone said that it is tiring to constantly listen to the physical explanation model that involves Rayleigh scattering when one instead should call things by their name - that the upper part of the atmosphere simply is blue because it has a blue color.
Is there a reason why they want to complicate things more than necessary? That a blue atmosphere not necessarily needs to be due to the fact that it is oxygen? But since neither Mars nor Venus is blue as Earth, there seemed to be a waste of angled propaganda methods here. The situation will be different if we study Mars' atmosphere in detail. There are images taken from the Hubble Space Telescope that reveal that Mars has a blue atmosphere. It has already been proven that Mars can sometimes have a cloudy sky. The Viking Lander’s first pictures showed a blue sky - then it changed to a peculiar red sky. This indicates that they are restrictive with data showing a blue sky on another planet. Why are they? If Mars' sky is blue as here on Earth, what could be the consequences? Will people draw a parallel with Earth that Mars is like Earth? Do people think that Mars' atmosphere actually is rich in oxygen then, even though it can be explained as Rayleigh scattering?
As I mentioned earlier, the concentration of ozone gas in the Earth's atmosphere is 2-8 ppm. Is this enough for the sky to assume its blue color? After a bit of searching online, I found the site Oxidation Technologies LLC. Where ozone gas has been dissolved in jars filled with water (water dissolves ozone better than air and evens out the mixture). If what is written on the site is correct, a concentration of around 50 ppm is required before one barely can distinguish the blue. Later it can be read that the larger the water tank is the easier it is to see the ozone. These jars and cans have a width of around 6-30 cm. What happens if the concentration drops 25 times while the width of the water tank increases 130,000 times! So 2 ppm ozone in a 40 km wide can, does one see the blue ozone then??

IMAGE ©Oxidation Technologies LLC
answer to that question will probably be NO if we return to conventional school physics:— The ozone layer is far too thin for it to be visible, and if the sky was blue due to ozone gas, the sun would also look blue, instead of yellow. No, what makes the sky blue is due to Rayleigh scattering.
As for my own opinion, I think that the explanatory model that describes Rayleigh scattering seems far-fetched or unnecessarily complicated - the relationship with reflection seems subtle. Also, the Rayleigh scattering is at the same altitude as the ozone, if I'm right? But I may be wrong, maybe Rayleigh scattering is a reality and maybe it can be proven in some better way? Personally, I do not have a super argument that can explain the opposite, not yet in any case, but maybe something will appear in the future? Yes, we'll see, we'll see…
Earth bluish atmosphere by CEO of EE |
|
Id:
|
||||
(1 or 2 digits)
|
||||
|
EECOM10 Comment Function 1.0
Developed by B.Lindqvist © 2021 Essential Electronics |
||||
| by CEO of EE | • 2020-09-18 | |||
| The secretive Kvarntorp pile | ||||
This article about nuclear physics was already on my EBR page but because it's such an interesting topic, I think it deserves a place on Innews. | ||||
C O N T E N T
I was once asked: What is it like to work at a nuclear power plant? Then I replied that the largest “nuclear power plant” in Sweden is quite close to the city of Motala, more precisely east of the city of Kumla. You can go there and see with your own eyes what it might look like. This slag heap of “Rödfyr” or BAS (burnt alum shale), constitutes a significant landmark in the plains of Närke, located right next to the society Hällabrottet. It is now an industrial monument that is decorated with “Art on pile”, as well as a gym - there are many steps to the top... Have been there several times and studied various phenomena. The city Örebro is for me an old place for studying, so I am well acquainted with the surroundings.
The history of and what is going on at this artificial slag heap is there very few that has been interested in. I would like to say - only a moderate interest in nuclear physics should be enough for a number of issues comes to mind. What exactly is fission? What does it take to start a fission process? How dangerous is radioactivity? etc. etc.
 The BAS on the Kvarntorp pile is radioactive. It can be ascertained
if a suitable measuring instrument is used. The highest value I have measured is 2 µSv / hour. Then my radex instrument beeps a lot while the measurement
was going on. It is difficult to know where it radiates most. You have to have a little patience when looking for such a place. The area next to the driveway
is a hot tip. Over 3 µSv / hour can be obtained if you measure on unbroken alum shale. There are places around Kvarntorp where you can make such
measurements. Otherwise, it is quite difficult to find areas in other places in our country that are radioactive at all, if you disregard other shale deposits,
nuclear power plants, and their facilities or houses built of blue aerated concrete or lightweight concrete (an older type of house building materials)...
Tips are welcome. When you are on the pile, you see signs here and there that warn of a “hot area”. Something in the subsoil creates an intense heat
so that water vapor is formed and seeps up - it smokes and smells of alum shale. Untreated alum shale (also called oil shale) is a mineral that smells of
oil / petroleum, ie it is rich in petroleum type Kerogen.
The BAS on the Kvarntorp pile is radioactive. It can be ascertained
if a suitable measuring instrument is used. The highest value I have measured is 2 µSv / hour. Then my radex instrument beeps a lot while the measurement
was going on. It is difficult to know where it radiates most. You have to have a little patience when looking for such a place. The area next to the driveway
is a hot tip. Over 3 µSv / hour can be obtained if you measure on unbroken alum shale. There are places around Kvarntorp where you can make such
measurements. Otherwise, it is quite difficult to find areas in other places in our country that are radioactive at all, if you disregard other shale deposits,
nuclear power plants, and their facilities or houses built of blue aerated concrete or lightweight concrete (an older type of house building materials)...
Tips are welcome. When you are on the pile, you see signs here and there that warn of a “hot area”. Something in the subsoil creates an intense heat
so that water vapor is formed and seeps up - it smokes and smells of alum shale. Untreated alum shale (also called oil shale) is a mineral that smells of
oil / petroleum, ie it is rich in petroleum type Kerogen.BAS is a residual product after the combustion of alum shale. It has a high iron content, hence the red color. In the past, lime quarries were often built where there were alum slates. The slate was fired in limestone kilns to be able to produce burnt lime (calcium oxide) and the ash (BAS) remained. The burnt lime was then quenched with water - calcium hydroxide was the result and was thus the mortar of that time. In Kvarntorp (1942-1966) they did not burn alum shale but instead heated it (pyrolysis). In this way, the petroleum could be separated - fuel oil, petrol, LPG, sulfur, ammonia... Only 20% of the slate's weight could be recovered, the remaining 80% was deposited on the ash heap (the pile). The process gave rise to large emissions of sulfur-containing substances that plagued the environment. The chimneys were high but it did not help much. Forests and vegetation in the immediate area died, metal objects rusted and the smell could be felt from the operations in Kvarntorp many miles away.

IMAGE ©Sydnarkenytt

IMAGE ©Sydnarkenytt
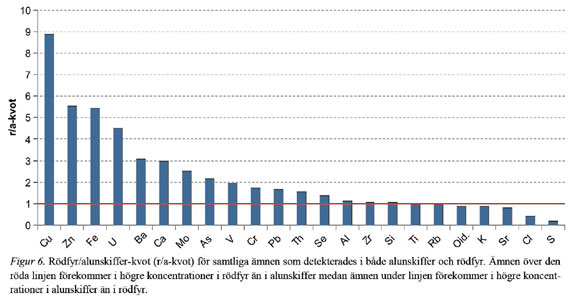
This is interesting (if true) because the uranium content exceeds 0.1%, which is the limit for what is considered “economically minable deposits”. We can make a simple calculation, to see how much natural uranium there is in the Kvarntorp pile. The total volume of the pile is 40 Mm³ (40 million cubic meters). The density of reasonably dry and packed BAS is 1.0 kg/dm³. The Kvarntorp pile uranium weight is then 1 × 40 • 10^6 × 0.00135 = 54,000 tonnes, in which case the content that applies to the BAS in Andrarum is taken into account. For the uranium content of 0.0375%, it will be 15,000 tons. Between 15,000 - 54,000 tonnes of uranium is thus found in the Kvarntorp-pile, or 0.38 - 1.35 kg per tonne of BAS-mass.
The next question one then asks is: What is the critical mass for BAS? Or rather: What is the critical mass for fresh BAS? The answer is that it is infinitely large - that no “critical mass” exists because the uranium is so diluted. Uranium is also not enriched, but it is natural, with a low content of U235. Especially when it comes to the Kvarntorp pile, the conditions were a bit special if you compare it with the way the lime kilns were operated. According to available information, it is remnants of oil that are still burned in the interior of the Kvarntorp pile. You can ask yourself how the air/oxygen gets into the glowing BAS? This problem/phenomenon with BAS that continues to burn for several decades does not exist in the piles around Sweden where the lime kilns built their BAS with. It is only in Kvarntorp that the burning of residual oil has taken place. If it were now the case that residues of oil/petroleum were still burning, a number of unburned residual products should be formed. We are talking here about combustion that takes place with a severe oxygen deficit. A lot of soot and black smoke should be generated. The stench from this type of combustion is almost unbearable. Despite this, it smells mostly of water vapor and has been filtered through alum shale. Then someone says: but the BAS over the hearth filters out all harmful substances. — Then I say: in that case, the Kvarntorp pile must be the world's most efficient oil combustion plant. The combustion just goes on and on without any human impact and without any harmful emissions happening...
What suggests that we are dealing with fission is the size of the reactor. The neutrons have plenty of time to split or be absorbed during their journey. The neutron utilization rate is extremely high here. The reactor also does not contain any control rods or boron. Before the BAS was deposited on the mound, it was very hot and it continued to be hot for a very long time. Accidents occurred when tractors got stuck on the pile. It was impossible to save the driver. It had to burn up - there was apparently no other alternative. I do not think the limestone oven makers had removed the alum slate that was still burning and was red hot, right? I have no information about why an alum slate that has undergone pyrolysis is so difficult to burn out. What could not be recovered from the pyrolysis a steam boiler was fired with and which also produced the necessary electric power, from a steam turbine plus its generator. Ash with a temperature of 600 - 700°C (1100 - 1300°F) should one be able to produce steam and electricity with too, but they chose to tip it on the pile instead.
The Kvarntorp pile is large in comparison with other BAS piles. If the other piles were as big as the Kvarntorp pile, would fission occur then? I read somewhere that they had tried to push pipes through the pile. The pipes would then be connected and it would then be possible to heat district heating water with the help of the heat generated from the BAS. It apparently did not work because the pipes melted due to the heat... It should be possible to read about this in the local newspaper, but then one has to know when it happened. Tips are gratefully received! Approximately every five years, heat formation causes cavities and the roof of the pile risks collapsing. However, this is routine and there are and always have been staff who take care of the problem. Excavators take up formed cavities and the remedy is spelled water, more water, clay, and Absol (a fire-fighting agent). If there are works of art in the way, they must be temporarily moved away while the extinguishing work is in progress. This happens from time to time. When I visited the pile in the autumn of 2020, I noticed that the steam formation had decreased in comparison with other visits. Maybe all the firefighting over the years finally has paid off...




If we are talking fission, which I believe, the fast neutrons will be absorbed by U238, which is converted to Pu239 and/or Pu241 and which can then be split by slow neutrons. This always happens and provides a significant energy supplement in all uranium reactors. If we instead play with the idea that there is combustion going on inside the pile (as “they” claim) then this heat development should spread upwards and finally, the whole pile should catch fire and burn like a bonfire. BAS has also been used as a base on football and tennis courts, with the result that less snow remains. Whether this is due to heat or something else I leave unsaid. I myself have scooped up 10 liters of BAS in a bucket without being able to establish any hint of heat generation.
Further back in time, there was more smoke from the Kvarntorp pile. Is it just me who thinks it's reminiscent of a volcano? The world's largest artificial volcano!

IMAGE ©Wikimedia Commons

IMAGE ©Creative Commons Attribution 2.5 se
In 1953, AB Atomenergi (AE) started up its new uranium extraction plant at Kvarntorp. They wanted the uranium in the alum shale. Before one can engage in enrichment and suchlike, uranium must be separated from all other substances and it is not entirely easy. However, it is only about classical chemistry to be able to obtain pure uranium. Already in Kvarntorp, the slate/culm was crushed and grounded and a first bearish separation was carried out. The uranium mass was then transported to Stockholm (Liljeholmen). At that time, all further uranium processing was carried out in densely populated areas. Two reactors (R1 and R3) were also located in this capital, but they did not care. It had not yet been realized that it required intimidation propaganda to stop people going for uranium. According to Galen Winsor, this is because - you do not want a large number of small units that produce electricity around the country, because then the state loses control (read: tax revenue) over electricity distribution and/or heat distribution (district heating). The uncontrolled spread of plutonium is another reason. Exactly what has happened regarding AB Atomenergi's operations in the 1940s and 1950s is it difficult to get an overall picture of. There were rumors that it was good practice for those involved not to talk about this. Then we have Ranstad in Västergötland where it was decided to mine and extract a maximum of 300,000 tons of uranium. However, it was only 213 tonnes, as it was eventually discovered that it was cheaper to import uranium from other countries.
The previously mentioned existence of natural reactors (oklo) tickles my imagination. What if liquid red hot magma is a kind of meltdown that takes place deep in the ground? Thus, real volcanoes, even those driven by fission processes in the underground. These are actually not crazy ideas but by many accepted hypotheses. There are several examples of educated people who think about these oddities. The general hypothesis why it is hot inside the Earth has nothing to do with the high pressure (due to gravity) but it is because when the Earth was formed it was a glowing ball that eventually cooled on the surface but inside it was still very hot, despite the fact that several billion years have passed. So, then we have come so far that it is time to step into the Venus sphere - without there being any connection between the Kvarntorp pile and Venus, or are there?
Is there life on Venus? - No it's too hot! It's so hot that lead melts. When you ask why it is so hot on Venus, you can get different answers: It is the carbon dioxide concentration that has become too high, and then the temperature has skyrocketed (the same scenario as we have to wait here on earth according to the same spokespersons). Or - it's the volcanoes on Venus that make it so hot. Since then, I have not heard any more explanation, because it can hardly be the thick cloud cover that causes the heat. As you know, clouds shield the sun's rays so that it gets colder. This means that it should be cold on Venus, but since Venus is closer to the sun, the stronger solar radiation should compensate for the cooling effect of the clouds. The result should be about the same as on Earth - quite comfortable...
On Mars, there are also clouds but quite a few. However, Mars is further away from the Sun than Earth is, so what will be the result? That it is quite pleasant temperatures and mostly clear blue skies over Mars. Mars' atmosphere is not as dense as Earth's either, so on Mars, you can count on a maximum utilization of what the Sun has to offer and not least - so that it can be tolerable living conditions. However, I am here on Earth where it is far enough to the sun and just enough clouds, ie tolerable living conditions. Everything becomes so simple when you think logically and rationally...
Mid-September 2020: It is very rare to hear anything about an expedition to Venus. It's a bit funny because while I was writing this text, a new discovery was published about Venus that had to do with the gas phosphine. That the atmosphere is gossiping that there might be some kind of life on Venus, but just maybe. It is usually formulated in that way so that they do not promise something that later did not turn out to be true. Because when some time has passed: they usually have a slighter different explanation... Sometimes one apparently timing coincidences one does not usually succeed with.
IMAGE ©Kumla kommun
 So a few months later, I discovered that a former fire chief named Stig Wikström wrote a book about the Kvarntorp pile and
which he published 2020-09-08 - ten days before this page was posted! Another coincidence that I had no idea about... I have not read the book
yet but from newspaper articles it says that the pile was more active in the past (the 70s, 80s, 90s) than today. From 1982, the Kumla fire brigade was responsible
for fires on the pile. In the 80's it often burned and sometimes a crater was formed due to the heat. On the walls of the crater one could see how the BAS glowed
(shone red). Expressions such as “fire” or “the pile is burning” are incorrect formulations according to Stig, but it is about a “chemical reaction”. If this
is correct, you ask yourself (especially if you are interested in chemistry) what kind of chemical reaction is it??? It is thus a chemical
reaction that creates an intense heat so that the remaining stubble is gasified and forms toxic flue gases, containing sulfur among other things, if I
understood correctly?
So a few months later, I discovered that a former fire chief named Stig Wikström wrote a book about the Kvarntorp pile and
which he published 2020-09-08 - ten days before this page was posted! Another coincidence that I had no idea about... I have not read the book
yet but from newspaper articles it says that the pile was more active in the past (the 70s, 80s, 90s) than today. From 1982, the Kumla fire brigade was responsible
for fires on the pile. In the 80's it often burned and sometimes a crater was formed due to the heat. On the walls of the crater one could see how the BAS glowed
(shone red). Expressions such as “fire” or “the pile is burning” are incorrect formulations according to Stig, but it is about a “chemical reaction”. If this
is correct, you ask yourself (especially if you are interested in chemistry) what kind of chemical reaction is it??? It is thus a chemical
reaction that creates an intense heat so that the remaining stubble is gasified and forms toxic flue gases, containing sulfur among other things, if I
understood correctly?
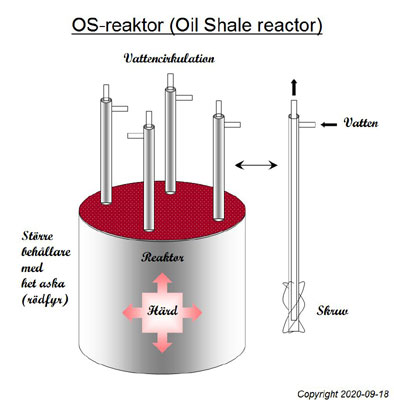
why not try if it works on a smaller scale... It was known that the ash was hot when it was placed on the pile, and then remained hot, yes for how long? You should therefore start by performing pyrolysis of alum shale, then deposit the hot ash in a container/reactor and continue until it is full. Thus, you have a container that will generate heat energy for several years, right? The simplest is to equip the reactor with the possibility of heating water, for heating residential... To keep the fission going, one can try watering the ashes from time to time.
Another suggestion is to fill a container with old BAS from Andrarum and then heat it up. To facilitate nuclear fission, the BAS should be mixed with water, ie a moderator. If the container is waterproof, a significant part of the mass may consist of water. Heating can be done by pushing in water vapor. And maybe a neutron source is also needed to kick start the fission process?
The way in which all piping is to be carried out, I leave to everyone to figure out. The most important thing is the BAS characteristics when it acts as a fuel and how it should be prepared, the rest always resolves, in some way...
Since I have not seen anyone else think in these terms, I am probably the author until further notice, if one disregards the research that AB Atomenergi carried out in the 40s and 50s, behind the curtain, however. I call it the OS reactor. It is a reactor whose fuel is “Oil Shale”. There is a great risk that nothing will happen because the reactor will be so small in comparison with the Kvarntorp pile. So, the only thing that is missing is to try to find out how it is with that thing...
There are two aspects regarding the business that was started in Kvarntorp: From an environmental point of view, it was a very bad idea to start mining alum shale to be able to produce oil, but the new knowledge about nuclear physics and nuclear power, which was acquired, was of course highly essential for society's future energy supply.
info about radioactivity
March 2022: Collection of information via anonymous source has resulted in notification of measured values. It has emerged that the ionizing effect from the ground on Kvarntorpshogen (KH) is higher than in the surroundings around the wrecked Chernobyl reactor in Ukraine, if one disregards hot spots. These hot sources are remnants of the graphite moderator which also spreads out, otherwise the main radiation source around CB is Cesium 137.Measured values around CB are 60-80 mR/h, while measured values of KH are 200-300 mR/h. The “Roentgen” unit was used on all instruments in the past. R measures the power [J/s] of the radiation - not the way ionizing radiation acts on living cells. Newer instruments measure absorbed radiation in “Sievert”, where the types of radiation also are weighted. Measuring with Sv means that you do not know what it is that radiates. As an example, many hazardous radioactive substances emit a lot of beta radiation, so to be able to measure it, special detectors are required. The same applies to alpha and gamma radiation, if we ignore neutron radiation. People who are nerdier in the field than me thus have a lot of special knowledge about how to proceed, which has then contributed to new detailed information about KH. Quote: “think your values are very low compared to what I measured”.
If we assume that fission processes are underway at KH, you can compare it with a nuclear power reactor, and then everything changes. Suddenly we have a collection of radioactive substances or fission products! The process at KH is so intense that it glows and smokes and has done so for a very long time - this does not happen at CB. It is as if a quantity of burnt-out uranium fuel rods from a nuclear power plant were thrown into a pile and covered with a layer of dirt, then alow people to stay there without any restrictions or warnings. It is also quite disadvantageous from a weather point of view. Precipitation leaches and transports radioactive substances to the groundwater. So do winds, which when it is dry outside scatter particles far and wide and then we have only talked about KH, not about other BAS-piles in Sweden. Finally: if what I wrote here is true, then the society Hallabrottet should be abandoned in the same way as the ghost town Prypjat, right?


The secretive Kvarntorp pile by CEO of EE |
|
Id:
|
||||
(1 or 2 digits)
|
||||
|
EECOM10 Comment Function 1.0
Developed by B.Lindqvist © 2021 Essential Electronics |
||||
| by CEO of EE | • 2012-08-16 | |||
| Mars - the red planet | ||||
This report about Mars was previously available in the News Bulletin. With the advent of Innews, it fits better here. This will be the first article and it also reflects the type of content that will appear here in the future. Keep in mind that this was not published yesterday, but the info has the same dignity now as then. It took a long time to fix a working comment function. After some effort, several PHP files emerged. So this is a little more advanced than the simpler older version, which is still used as a guestbook etc. First I had to read a number of books about Javascript and PHP programming to understand the principle. I have avoided using external code packages / modules such as Jquery, JSON, and Ajax - everything is based on self-constructed code - text files act like databases, and so on... | ||||



I find it interesting that NASA regularly sends spacecraft to various planets but are unsure about the reports really is true? I actually think that humans have visited the Moon and even landing on Mars and Venus with various widgets but as I said; unsure if it have taken place after the premises it so to speak have been published.
I do not think they visit the moon with the technology that was available 1969. NASA did this feat six times without that any serious technical error occurred during the landing and the take-off. If one compare the amount of fuel that was required to remove themselves from the earth seems it unreasonable that with a relatively small craft both land and take off from the Moon, but everything went smoothly, however there arose errors in the cameras that were used from time to time - usually when something important was about to be photographed.

In fact, just the photography has been a dilemma from Mercury to the latest spaceflight. The first time they came to Mars and was about to photograph its surface from space with the probe Mariner 9, so was it not possible because the planet was enveloped in a sandstorm - a global sandstorm - it happened again in 2001 and was photographed with the Hubble Space Telescope. Scientists do not know how this is possible considering that the atmospheric density only is a fraction of that on Earth. The gravity on Mars is much smaller than on Earth when the planet is about half of Earth's size. Despite this is the land area the same as the Earth's land area.


Other photographs from Hubble show that Mars houses both bluish atmosphere and clouds. This talks it only little about - it feels like clouds on Mars sky not can be relevant, but there it is. Even dense clouds and fog have been observed from orbiters around the planet.

The first photographs taken by the Viking Lander confirmed a clear blue sky - future pictures show a reddish sky. These pictures looks suspicious because the landscape seems to be sterile and dead, thereto is the red shade obvious, well almost artificial unnatural.




One gets evidence for this when the images are balanced with Photoshop - the sky turn blue and the scenery gets a realistic expression. Even the black parts or the frame that surrounds the pictures have not managed to avoid being exposed from the red-applied paint effect. Just the fact that the black areas/edges are brownish proves that the entire image has been distorted before it has being released into the public spotlight. This is not only one image, it is many. So the conclusion to be drawn is that NASA wants that it shall be red on Mars. It is only clouds and the polar regions which are white, the rest of Mars is red!
 The encircled is the atmosphere
The encircled is the atmosphereWhen looking at the pictures from the Hubble Space Telescope we see hints of dark brown and green (watch the images from the Hubble above). One may wonder why NASA does not edit these pictures? Even a bluish atmosphere is visible as the one we have here on earth. A blue color indicates either oxygen or nitrogen or both - these elements creates a bluish atmosphere. When in contrast, a probe take pictures on March while it is in an orbit, one never see a shred of either blue or a cloudy sky.


The close-ups of March surface that was taken with equipments from MSSS are all black and white, although the images strongly reminiscent of vegetation. NASA and MSSS have therefore refrained from using color pictures on these interesting surface textures (at this alien planet), which are similar to trees or shrubs and which cast shadows over the landscape. It is noteworthy, as an orbiter not need to link the color data from a ground-placed source.
Even Arthur C. Clarke questioned to what this really is about, but he got no comprehensive answer. Instead responded NASA with a repetition, ie a new photograph of the site (the left black and white picture above) - which then showed a barren reddish desert landscape again. Several researchers have studied the detailed images from MGS and argued that it is sufficient proof for (at least) vegetation on Mars, but NASA always knows best and dismisses these assumptions without further notice. Some also claim they can discern herds of earth-like animals on the Mars surface.
These astoundingly ground structures are located in adjacent of the polar regions, but it has never hit anyone (at NASA) that it is exactly there they should put the resources, if the search for life is the point. No, instead they focus every expedition onto stone deserts around the Mars equator, which mostly can be compared with Sahara or the Gibson Desert in Australia.

Pictures taken on Mars are generally unnatural or poorly colored - this should be a good view?
One can now rest assured that the blunders by the clumsy red-imposed images are corrected by NASA. It would be remarkable if one could balance images from Curiosity so that the sky turns blue again - there is probably no risk. We simply have to realize that nothing beyond what we've already seen will ever appear and that other landing areas than inanimate Mars deserts never become relevant to land on - even in the future.
Mars - the red planet by CEO of EE Link image and all other images: ©NASA |
|
Id:
|
||||
(1 or 2 digits)
|
||||
|
EECOM10 Comment Function 1.0
Developed by B.Lindqvist © 2021 Essential Electronics |
||||






